|
NEURONAL SUPPORT CELLS OF THE MALE SENSILLA - RAYS
 Click pictures for new window with figure and legend Click pictures for new window with figure and legend
1 Ray Composition and Structure
Nine bilateral pairs of finger-like rays (numbered (n) 1 to 9, Left/Right) radiate from the tail and are embedded in a cuticular fan (MaleRayFIG 1). In male mating behavior the rays are required for initial contact response upon encountering the hermaphrodite and for turning during the male's search for the vulva (MaleNeuroFIG 3; Neuronal Support Cells of the Male - Overview; Liu and Sternberg, 1995; reviewed in WormBook: Male Mating Behavior - Barr and Garcia).
MaleRayFIG 1: Male sensory rays. A. Nomarski DIC of adult male tail, ventral view. B. Illustration showing detail of boxed region in A as a "cut-away" view.
All rays have the same cellular composition and general organization (MaleRayFIG 1B). Each contains sensory dendrites of an A-type (RnA) and B-type (RnB) neuron (MaleRayTABLE 1). Neuron processes are surrounded by a tube formed by the ray's single interfacial cell, the ray structural cell (Rnst) (MaleRayFIG 4A-C; Sulston and Horvitz, 1977; Sulston et al., 1980). The Rnst, in turn, is ensheathed by hyp7 and by cuticle. Except for ray 6, all rays are open at their tip. This opening is visible by DIC optics as a "ring-and-dot" formed by the hyp and Rnst encircling the RnB tip (MaleRayFIG 1B, MaleRayFIG 6). Despite being exposed to the environment, RnB neurons do not dye-fill with FITC or DiO (Perkins et al., 1986). The RnA, RnB and Rnst cells of a single ray are closely related by lineage, arising from a common great-grandmother precursor cell, Rn (MaleRayFIG 2).
MaleRayTABLE 1: Summary of ray cells. n = rays 1-9.Rn = V5/V6 /T.xxxxx in the male. R1 derives from V5, R2-6 from V6 and R7-9 from T.
MaleRayFIG 2: The ray. A. Diagram showing nuclei in left lumbar ganglia, lateral view, left side. Ray cell body positions are variable. Unmarked nuclei correspond to juvenile or hermaphrodite cells. (Adapted from Sulston et al., 1980.) B. Ray sublineage showing cell divisions with time. Hours are post-hatching at 20°C. C. Nomarski DIC with illustration showing ray cell trajectories in the adult ray, lateral view. (Rn) Ray precursor cell; (RnA and RnB) ray neurons; (Rnst) ray structural cell; (PAG) pre-anal ganglion. n=rays 1-9. Ray cells of each side are located together in their respective left or right lumbar ganglia (LGL/R) (MaleRayFIG 2 and 3A). Cell body positions within the ganglia are variable. Except for R6BL/R, RnB neurons have large cell bodies that contain dilated cisternae (MaleRayFIG 3B). Ray neurons (but not the Rnsts) send axons, via commissures, from the LGL/R to the pre-anal ganglion (PAG) (MaleRayFIG 2; MaleRayFIG 4; Sulston et al., 1980).
MaleRayFIG 3: Ultrastructure of the lumbar ganglia. A. Low-power TEM highlighting the lumbar region with the rays, transverse section. (Image source: N2Y [MRC] 27.) B. Higher magnification TEM of the ray cell bodies within the lumbar ganglia from boxed region of A, transverse view. (N) Nucleus; (PhsolL/R) Phasmid socket cells. (Image source: N2Y [MRC] 1350.)
|
MaleRayFIG 4: Epifluorescent images of ray neurons and structural cells. A. Transgenic animals expressing the ram-5::GFP reporter gene in the adult ray, ventral view. (Strain source: R.Y.L. Yu and K.L. Chow.) B. Dorsal view. C. Transgenic animals expressing the flp-6::GFP reporter gene, ventral view. (Strain source: K. Kim and C. Li.)
The Rnst cell functions both as a sheath and a socket cell. Like the sheath cell, the Rnst surrounds neuron dendrites and contains numerous vesicles that presumably secrete material into the ray lumen. Like the socket cell, the Rnst acts as a transitional cell linking the sense organ to the surrounding hyp via adherens junctions (MaleRayFIG 5 and MaleRayFIG 6).
MaleRayFIG 5A&B: Longitudinal views of male rays. A. TEM of cross section of ray 2. (Image source: MaleRays [MRC] 747_13.) B. Diagram showing longitudinal section of ray 2 shown in A. (AJ) Adherens junction; (Hd) hemidesmosomes; (hyp) hypodermis. (Adapted from Sulston et al., 1980.)
MaleRayFIG 5C&D: Transverse views of male rays. A. TEM of cross section of ray 2, transverse view. (Image source: MaleRays [MRC] 747_19.) B. TEM of cross section of ray 5, transverse view. (Image source: MaleRays [MRC] 747_15.) (AJ) Adherens junction; (Hd) hemidesmosomes; (hyp) hypodermis.
2 Ray Development
Rays are generated post-embryonically from the most posterior L1 lateral seam cells of each side, V5 (L/R), V6 (L/R) and T (L/R) (see Male Epithelial System - Introduction). The characteristic finger-like structure of the ray is generated by a process of morphogenesis whereby part of each ray cell remains attached to the body wall while surrounding hyp and body wall tissues migrate anteriorly (summarized in MaleHypFIG 6A; see Male Epithelial System - Introduction for details). Interactions between the Rnst and hyp are thought to play an important role in ray morphogenesis. Loss of function mutations in several genes cause a lumpy ray phenotype or disrupt ray extension (a Ram phenotype - Ray abnormal morphology or a Mab phenotype - Male abnormal). Characterization of one such gene, ram-5, reveals that it encodes a novel protein that is expressed in the Rnst cells and may act in a glycosylation-dependent pathway that regulates hyp and Rnst cell-cell interactions (Yu et al., 2000; Ko and Chow, 2003).
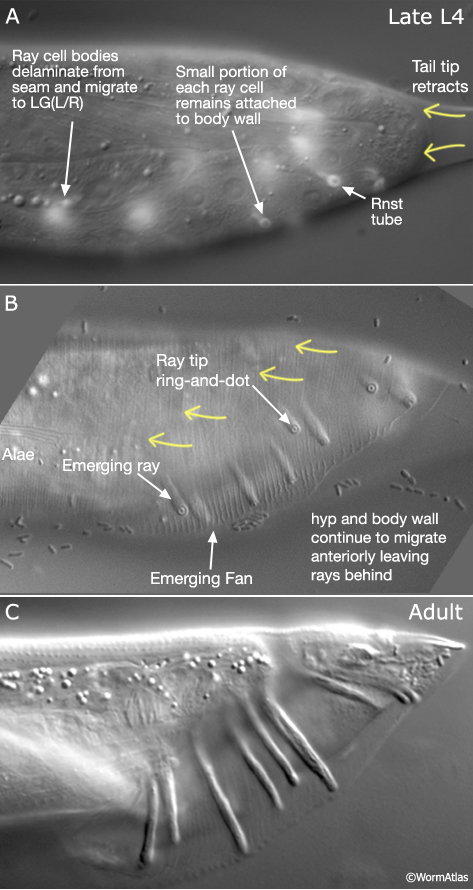
3 Ray Identity
The Rnst is also thought to play a role in defining ray-specific morphology. Each left/right ray pair has a characteristic shape and opens on a particular side of the fan (MaleRayFIG 1A ; MaleRayTABLE 2). These morphological attributes, expressed along with distinct combinations with neuronal characteristics (e.g. neurotransmitter fate, axons trajectory, chemosensory receptor), give each ray pair a unique identity (Sulston and Horvitz, 1977; Loer and Kenyon, 1993; Troemel et al., 1995; Lints et al., 2004; L. Jia and S.W. Emmons, pers. com.). Ray identities may reflect functional specialization of rays in aspects of mating behavior. For example, dorsally-opening rays (rays 1, 5, and 7) are required for contact response during a dorsal encounter with a hermaphrodite; ventrally-opening rays (rays 2, 4, and 8) function with other ventral sense organs in ventral encounters; the posterior T rays (7-9) are required for turning (Liu and Sternberg, 1995). Ray identities are specified by regulators (including Hox genes, a TGF-beta signal, DM-domain and Pax-6 transcription factors) that act in distinct combinations in each ray pair (Chow and Emmons, 1994; Savage et al. 1996, Zhang and Emmons, 1995; Lints and Emmons, 1999; Lints and Emmons, 2002; reviewed in WormBook: Male Development - Emmons).
|
MaleRayTABLE 2: Summary of ray identity characteristics.
* may be defined by differentiated characteristics the Rnst cell. PKD-2 / LOV-1 expression: Barr and Sternberg, 1999. CWP-1, -2, -3 and -4: Portman and Emmons, 2004. DA (dopamine): Sulston and Horvitz, 1977. 5HT (serotonin): Loer and Kenyon, 1993, Lints et al., 2004. flp/FLP (FMRFamide-like-peptide reporters for flp-5, flp-6, flp-17 /antibody-positive): Lints et al., 2004.
4 Ray Neuron Connectivity
Early reconstruction data suggest that neurons of different rays may synapse with different combinations of targets (The Male Wiring Project). The data also suggest that there may be a high degree of connectivity between neurons of certain rays (MaleRayFIG 7; see Individual neurons). As observed in the post-cloacal sensilla, some rays (e.g. R3AL) may be motor as well as sensory neurons because they form neuromuscular junctions with sex muscles (MaleRayFIG 8; Male Muscle System - Introduction).
MaleRayFIG 7: Connectivity between ray neurons. TEM showing synapses between different ray neurons, transverse section. (Image source: N2Y [MRC] PAG 522.) (vBMW) Ventral body wall muscle; (PAG) pre-anal ganglion.
MaleRayFIG 8: Neuromuscular junctions within the rays. TEM of featuring neuromuscular junction between rays and sex muscles, transverse view. (Image source: N2Y [MRC] L849.) (polL) Posterior outer longitudinal; (dglL) diagonal muscle; (BWM) body wall muscle.
5 List of Male Ray Cells
6 References
Barr, M.M. and Sternberg, P.W. 1999. A polycystic kidney-disease gene homologue required for male mating behaviour in C. elegans. Nature 401: 339-40. Abstract
Chow, K.L. and Emmons, S.W. 1994. HOM-C/Hox genes and four interacting loci determine the morphogenetic properties of single cells in the nematode male tail. Development 120: 2579-92. Article
Ko, F.C. and Chow, K.L. 2003. A mutation at the start codon defines the differential requirement of dpy-11 in Caenorhabditis elegans body hypodermis and male tail. Biochem. Biophys. Res. Commun. 309: 201-8. Abstract
Lints, R. and Emmons, S.W. 1999. Patterning of dopaminergic neurotransmitter identity among Caenorhabditis elegans ray sensory neurons by a TGFbeta family signaling pathway and a Hox gene. Development 126: 5819-31. Article
Lints, R. and Emmons, S.W. 2002. Regulation of sex-specific differentiation and mating behavior in C. elegans by a new member of the DM domain transcription factor family. Genes Dev. 16: 2322-6. Article
Lints, R., Jia, L., Kim, K., Li, C. and Emmons, S.W. 2004. Axial patterning of C. elegans male sensilla identities by selector genes. Dev. Biol. 269: 137-51. Article
Liu, K.S. and Sternberg, P.W. 1995. Sensory regulation of male mating behavior in Caenorhabditis elegans. Neuron 14: 79-89. Article
Loer, C.M. and Kenyon, C.J. 1993. Serotonin-deficient mutants and male mating behavior in the nematode Caenorhabditis elegans. J. Neurosci. 13: 5407-17. Article
Perkins, L.A., Hedgecock, E.M., Thomson, J.N., Culotti, J.G. 1986. Mutant sensory cilia in the nematode C. elegans. Dev. Biol. 117: 456-487. Article
Portman, D.S. and Emmons, S.W. 2004. Identification of C. elegans sensory ray genes using whole-genome expression profiling. Dev. Biol. 270: 499-512. Article
Savage, C., Das, P., Finelli, A.L, Townsend, S.R., Sun, C.Y., Baird, S.E. and Padgett, R.W. 1996. Caenorhabditis elegans genes sma-2, sma-3, and sma-4 define a conserved family of transforming growth factor beta pathway components. Proc. Natl. Acad. Sci. USA 93: 790-4. Article
Sulston, J.E. and Horvitz, H. R. 1977. Post-embryonic cell lineages of the nematode Caenorhabditis elegans. Dev. Biol. 56: 110-156. Article
Sulston, J.E., Albertson, D.G. and Thomson, J.N. 1980. The Caenorhabditis elegans male: Postembryonic development of nongonadal structures. Dev Biol. 78: 542-576. Article
Sulston, J.E., Schierenberg, E., White J.G. and Thomson, J.N. 1983. The embryonic cell lineage of the nematode Caenorhabditis elegans. Dev. Biol. 100: 64-119. Article
Troemel, E.R., Chou, J.H., Dwyer, N.D., Colbert, H.A and Bargmann, C.I. 1995. Divergent seven transmembrane receptors are candidate chemosensory receptors in C. elegans. Cell 83: 207-18. Article
Yu, R.Y., Nguyen, C.Q., Hall, D.H. and Chow, K.L. 2000. Expression of ram-5 in the structural cell is required for sensory ray morphogenesis in Caenorhabditis elegans male tail. EMBO J. 19: 3542-55. Article
Zhang, Y. and Emmons, S.W.1995. Specification of sense-organ identity by a Caenorhabditis elegans Pax-6 homologue. Nature 377: 55-9. Abstract
|

This chapter should be cited as: Lints, R. and Hall, D.H. 2009. Male neuronal support cells, rays. In WormAtlas. doi:10.3908/wormatlas.2.10
Edited for the web by Laura A. Herndon. Last revision: July 17, 2013. |
|